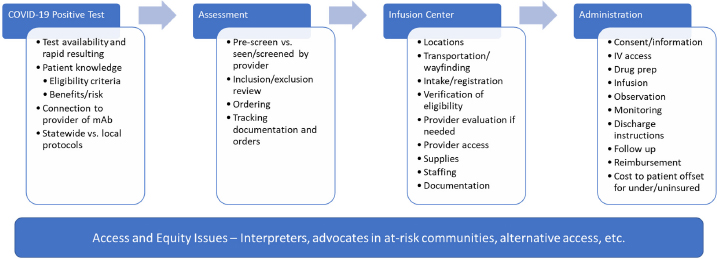
Monoclonal Antibody Treatments
The criteria to determine if you are eligible include the severity of. This amount is known as viral load.

Monoclonal And Polyclonal Antibodies In Covid 19 Treatment Mci Diagnostic Center Llc
Etesevimab also known as LY-CoV016 and LY3832479 is another neutralizing monoclonal antibody that binds to a different but overlapping epitope in the RBD of the SARS-CoV-2 S protein.
Monoclonal antibody treatments. For example some monoclonal antibodies mark cancer cells so that the immune. The antibodies signal to immune cells. On February 9 2021 the FDA issued an EUA for the investigational monoclonal antibody therapy bamlanivimab and etesevimab administered together for the treatment of mild-to-moderate COVID-19 in adults and pediatric patients with positive COVID-19 test results who are at high risk for progressing to severe COVID-19 andor hospitalization.
What is monoclonal antibody treatment. Monoclonal antibodies are synthetic antibodies that mimic the immune systems response to an infection. Brandon Webb MD infectious diseases says.
An injected monoclonal antibody seeks out cancer cell proteins. What is Monoclonal Antibody Treatment. It is important for people diagnosed with COVID-19 to ask their physician if monoclonal antibody treatment is right for them.
Monoclonal antibodies are laboratory-made proteins that mimic the immune systems ability to fight off harmful viruses and bacteria which can cause disease. This page displays monoclonal antibody therapeutic treatment shipment locations nationwide. Monoclonal antibodies are made in a lab and work similarly to antibodies your immune system makes to fight infection.
Monoclonal antibody therapy For people at high risk of getting very sick from COVID-19 monoclonal antibody treatment given early can significantly reduce the risk of progressing to severe COVID-19 disease and needing hospitalization. Learn more about targeted therapy. Bamlanivimab also known as LY-CoV555 and LY3819253 is a neutralizing monoclonal antibody that targets the RBD of the S protein of SARS-CoV-2.
Monoclonal antibodies are an important tool that help people diagnosed with COVID-19 treat the disease early and help prevent severe sickness. Some early evidence suggests that mAb treatment can reduce the amount of the SARS-CoV-2 virus the virus that causes COVID-19 in a persons system. Some monoclonal antibodies are also immunotherapy because they help turn the immune system against cancer.
83 Zeilen Monoclonal antibody therapy is a form of immunotherapy that uses monoclonal antibodies mAb to bind monospecifically to certain cells or proteins. Many monoclonal antibodies are used to treat cancer. Monoclonal antibodies MABs which trigger the immune system to treat cancer.
This page displays monoclonal antibody therapeutic treatment. Examples of MABS that work in this way include. The monoclonal antibody bind to the proteins.
They will ask you some screening questions and tell you about the treatment. Finding the right antigens for cancer cells is not always easy and so far mAbs have proven to be more useful against some cancers than others. Monoclonal antibody treatment helps your body fight COVID-19 while your immune system begins to make its own antibodies.
Our monoclonal antibody treatments of REGEN-COV are given by intravenous infusion through your veins. The antibody treatment must be given to a patients within seven days of symptoms and is most effective for first time COVID-19 patients. Immunotherapy with armed or unarmed monoclonal antibodies targeting tumour-specific antigens has emerged in the last two decades as a novel potential adjuvant treatment for all types of neoplasia.
Monoclonal antibodies are used to treat many diseases including some types of cancer. You will receive a phone call from a monoclonal antibody -trained pharmacist at Michigan Medicine if its possible that you are eligible. Initial data shows that mAb treatment reduces the risk of hospitalization and death in COVID-19 patients and slows down progression of the virus in the community.
- 2 - Who is eligible for treatment with monoclonal antibodies. Most effective on patients who do not have antibodies against COVID-19 to recall monoclonal antibodies mimic the bodies natural antibody response Dr. The objective is that this treatment will stimulate the patients immune system to attack those cells.
It should be given as soon as possible after a positive COVID-19 test or within ten days of developing symptoms. The immune cells arrive and punch holes in the cancer cell. Its an outpatient procedure for patients who tested positive for COVID-19 and are at high risk of developing a more serious case of coronavirus.
They are a type of targeted cancer therapy which means they are designed to interact with specific targets. In clinical studies monoclonal antibody treatments were shown to be safe and effective. Rituximab Mabthera a treatment.
Nevertheless results from ongoing Phase III clinical trials are encouraging as disease stabilisation. The treatment can also shorten the duration of COVID-19 symptoms. Many challenges to its implementation as a safe and viable therapy for brain tumours still need to be addressed.
Monoclonal antibodies or mAbs are made in a laboratory to fight a particular infectionin this case the virus that causes COVID-19and are given to patients directly with an infusion or a shot. The cancer cell dies. To make a monoclonal antibody researchers first have to identify the right antigen to attack.
This page displays monoclonal antibody therapeutic treatment shipment locations nationwide. What is a monoclonal antibody. Food and Drug Administration FDA has issued Emergency Use Authorizations EUAs permitting the use of the following mAb therapeutics for the treatment.
Thats why mAb treatment may help patients who are at high risk for severe symptoms or hospitalization. Currently there are two monoclonal antibody therapies approved for use under an Emergency Use Authorization. Its called monoclonal antibody mAb treatment.
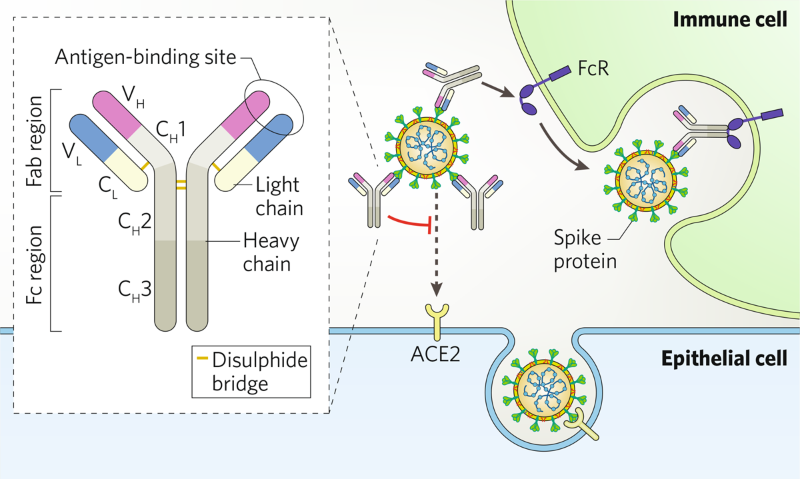
Hunting For Antibodies To Combat Covid 19

What Are Monoclonal Antibodies And Can They Treat Covid 19 Iav
What Is Monoclonal Antibody Therapy Who Is Eligible To Receive It Houston Methodist On Health

Mrna A Novel Avenue To Antibody Therapy Molecular Therapy

Rapid Expert Consultation On Allocating Covid 19 Monoclonal Antibody Therapies And Other Novel Therapeutics January 29 2021 Rapid Expert Consultation On Allocating Covid 19 Monoclonal Antibody Therapies And Other Novel Therapeutics January 29

Monoclonal Antibody Treatment Idph

Monoclonal Antibody Treatment Idph
Antibody Treatments For Covid 19 Are Worth The Effort Doctors Say Shots Health News Npr
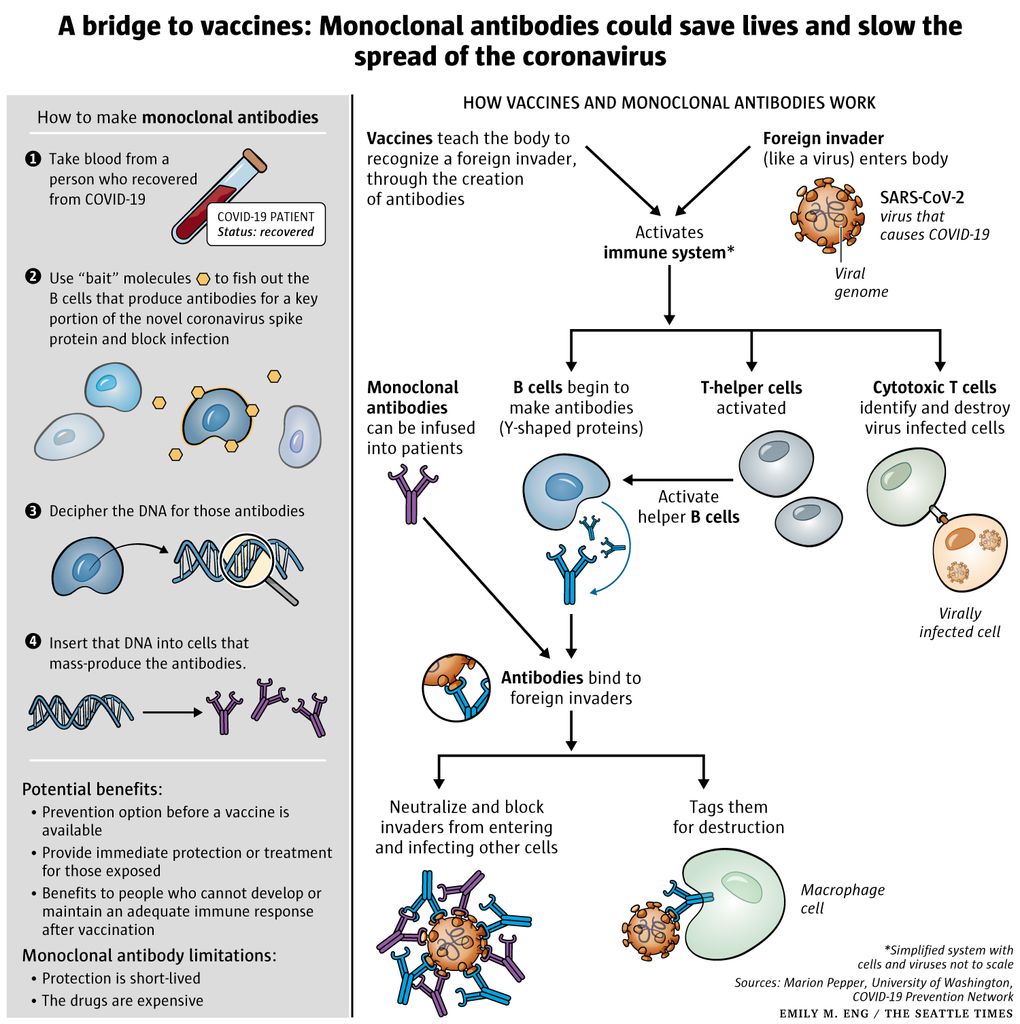
Monoclonal Antibodies Could Fill The Covid 19 Treatment Gap Until Vaccines Arrive But At A Cost The Seattle Times
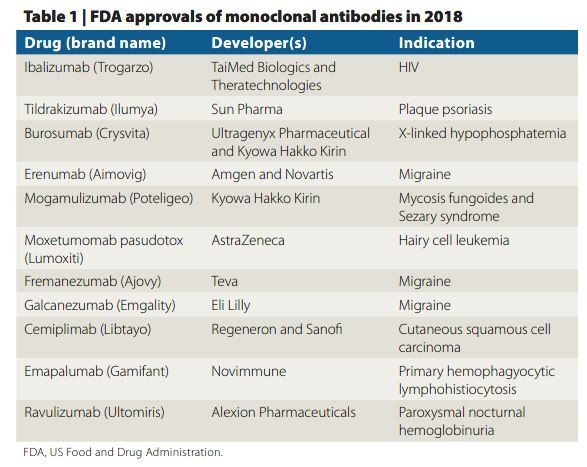
Moving Up With The Monoclonals
Monoclonal Antibodies And Other Novel Therapeutics In Covid 19 Treatment Mayo Clinic School Of Continuous Professional Development
Definition Of Monoclonal Antibody Nci Dictionary Of Cancer Terms National Cancer Institute
Johns Hopkins Helps Administer Covid 19 Antibody Therapy At Baltimore Convention Center Field Hospital Johns Hopkins Medicine

An Update On Covid 19 Treatments Monoclonal Antibodies Convalescent Plasma And Other Promising Developments Covid 19 Johns Hopkins Bloomberg School Of Public Health

/do0bihdskp9dy.cloudfront.net/08-13-2021/t_f202a13e50794e99b02ec78aa7ee1a02_name_file_1280x720_2000_v3_1_.jpg)


Post a Comment for "Monoclonal Antibody Treatments"